eta2-Imine complexes of zirconium, or zirconaaziridines, have attracted attention as amino carbanion equivalents. Insertion of unsaturated organic compounds into the polar Zr-C bonds of zirconaaziridines leads to amines, allylic amines, heterocycles, diamines, amino alcohols, amino amides, amino amidines, and amino acid esters.
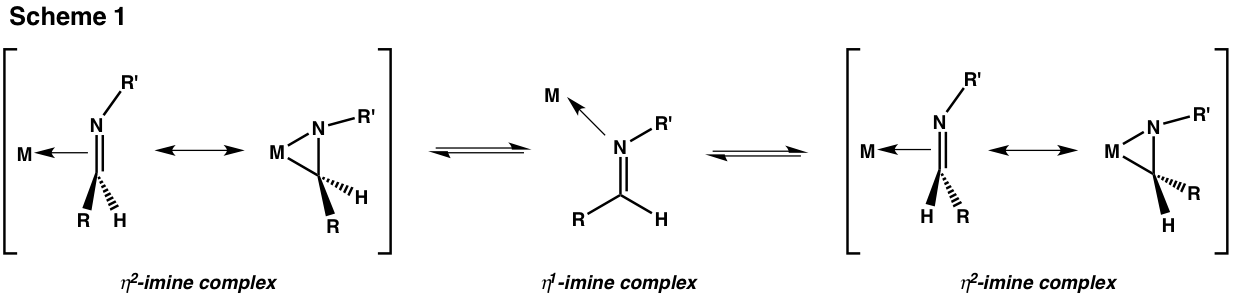
Zirconaaziridines contain a chiral center if their ring carbon bears two unequal substituents. That carbon is capable of configurational inversion if its eta2 isomer equilibrates with its eta1 analog (Scheme 1).
By treating a racemic zirconaaziridine 1 with a chiral electrophile (Scheme 2) under conditions where enantiomer interconversion is faster than electrophilic attack we have been able to achieve dynamic kinetic resolutions, i.e., to produce products enriched in one configuration of the carbon originally bound to Zr. We have found (R,R)-diphenylethylene carbonate ((R,R)-DPEC) to be an excellent chiral electrophile, giving zirconacycle 2 with de's up to 90%.
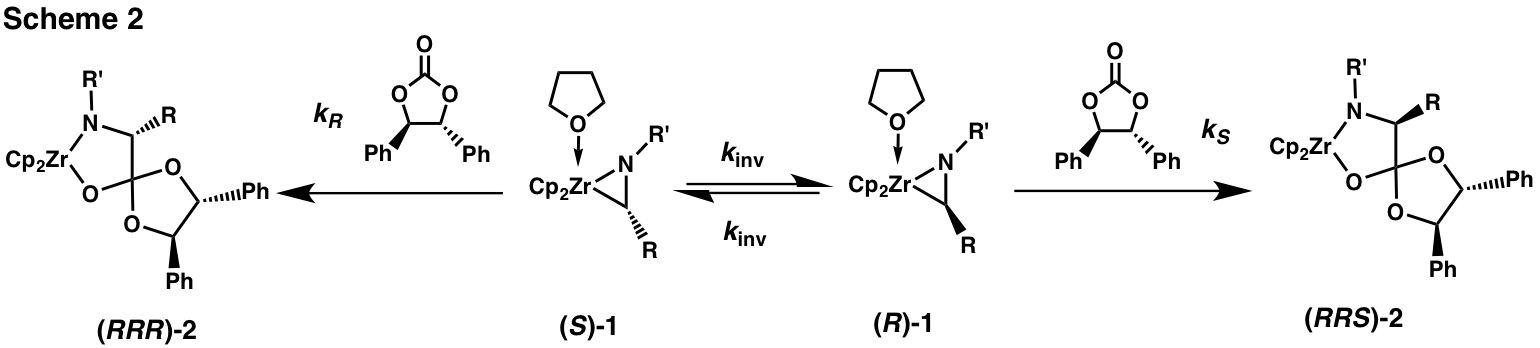
he stereochemistry of the new chiral center in 2 is determined by the competition between the (first order) interconversion of the enantiomers of 1 and the (second order) insertion of the carbonate into their Zr-C bonds (Scheme 2). When insertion is slow relative to the rate of enantiomer interconversion (the condition in eq 1, where Keq = 1) we will obtain a diastereomer ratio of kR/kS.
Achieving that maximum diastereomer ratio often requires slow addition, with a syringe pump, of the (R,R)-DPEC to the zirconaaziridine. This procedure fulfills the conditions of eq 1 by keeping [(R,R)-DPEC] low. However, for any given zirconaaziridine it is helpful to know the rate constant, kinv, for enantiomer interconversion. We have determined kinv in several cases, one fast (10 s 1), one slow (5.3 x 10-6 s 1) and one intermediate (9.1 x 10-3 s 1).
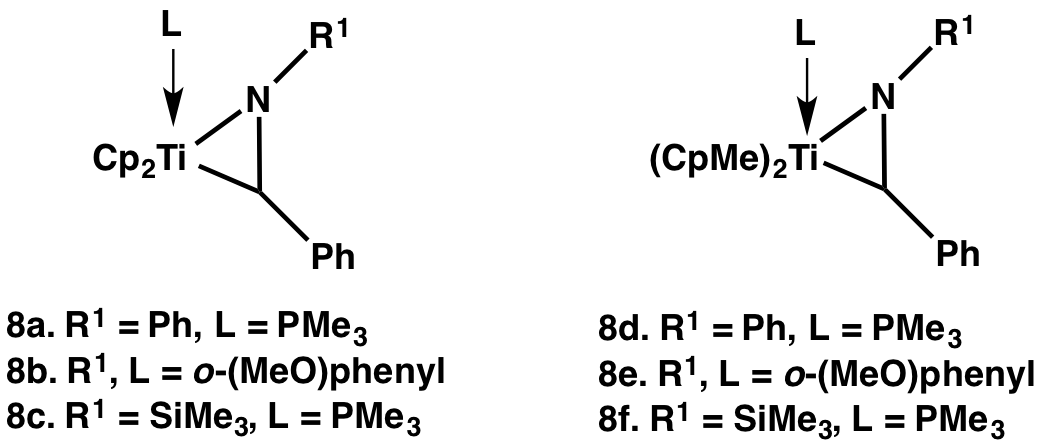
Motivated by the need for metallaaziridines with larger kinv, we have prepared a number of titanaaziridines, and have measured the rates by which their enantiomers interconvert by variable-temperature NMR.